Axia Lab – RFID & Supply Chain Testing Services
Is your organization facing supply chain issues or challenges?
We can customize testing to find answers.
At Axia Lab, our experienced researchers can help with customized testing protocols for RFID testing and validation, as well as waste valorization, smart packaging and much more. Learn more about RFID Validation below or visit our Order Testing page to create a customized testing request.

RFID Validation
Axia is a neutral-body, and its RFID Testing services are performed in partnership with experts from MSU’s College of Engineering and School of Packaging. All tests are conducted at the Axia Lab which is located at the MSU St. Andrews facility in Midland, MI.
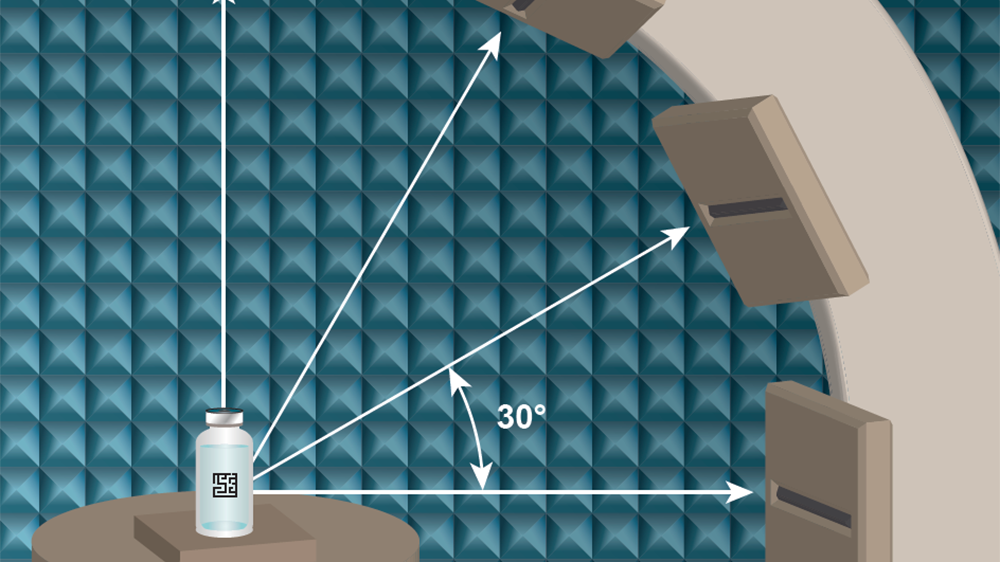
Axia Lab provides third-party validation services for Radio Frequency Identification (RFID) tagging. Samples received by Axia’s team will be subjected to a series of standardized and tailored robust tests based on the GS1 standards for RFID tags.
The Axia team not only tests RFID tags, but can conduct tag testing on the devices and/or medication they are intended to track, and offers services for both solution providers and end users. Axia Lab is licensed to receive controlled substances schedule 2-5.
Our deep expertise in RFID technology and its application across industries is our key differentiator. As RFID tags are being used for tracking even more types of products, Axia Lab is expanding testing services as well, including validation and testing for pharmaceuticals, food packaging, consumer goods, and other markets.
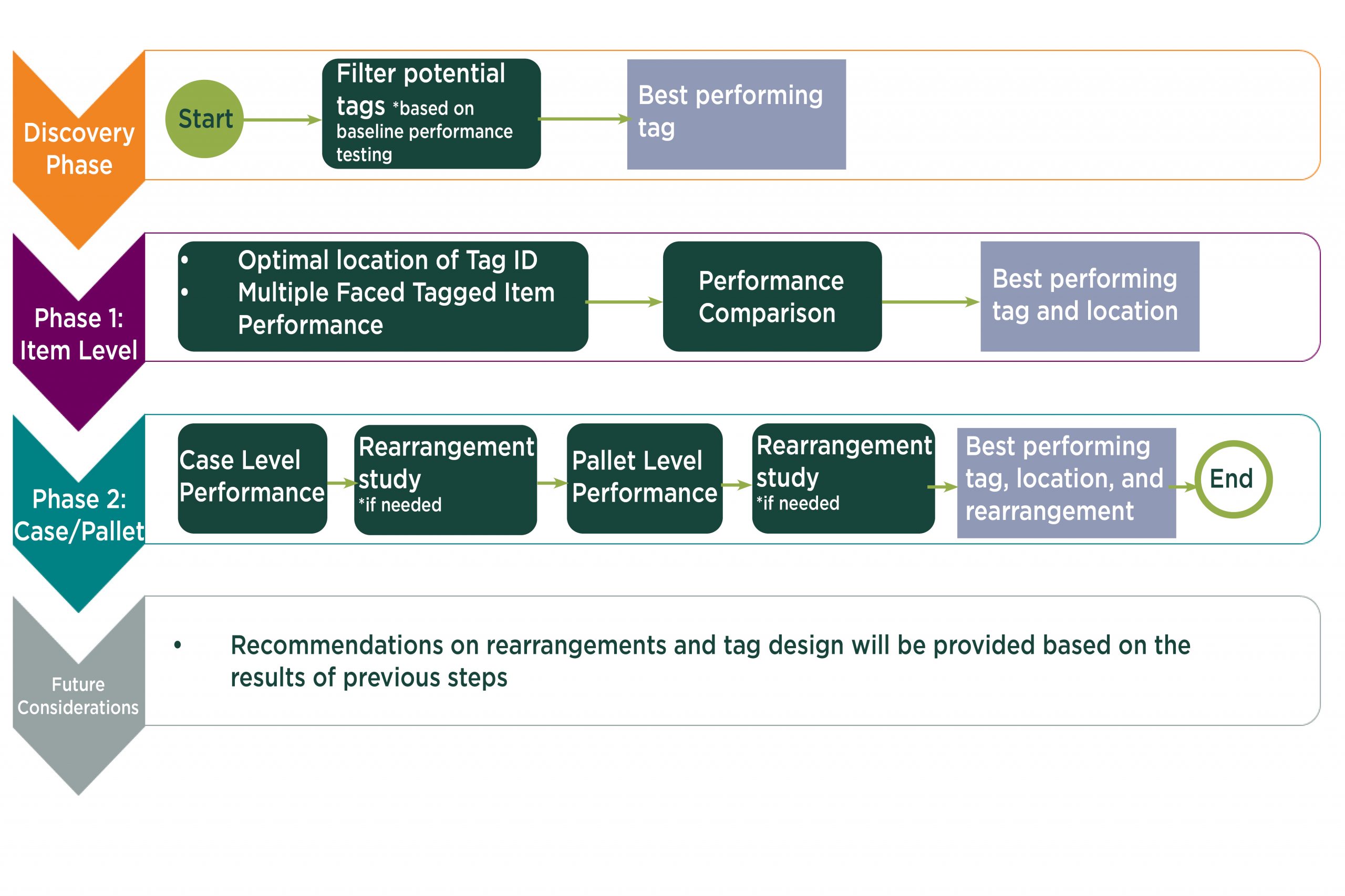
Axia’s Process
Once the top candidates have been identified, Axia will process to the case and pallet level readability testing to evaluate readability or packaging rearrangement to maximize readability.
The Lab has the capacity to do the above sequence for multiple SKUs as they vary in physical dimension.